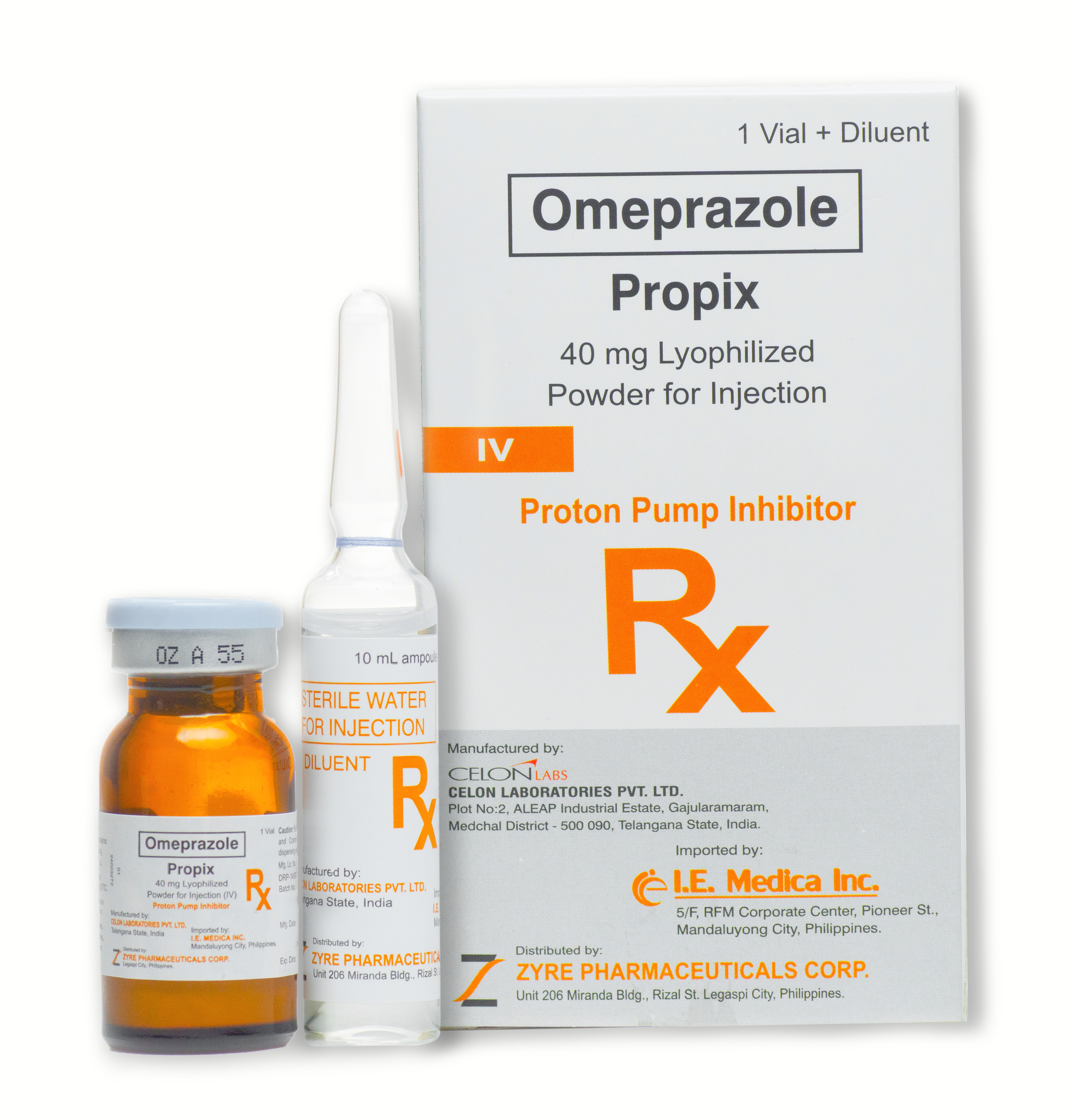
PROPIX
40 mg Lyophilized Powder for Injection (IV)
Omeprazole belongs to a class of antisecretory compounds, known as ‘proton pump inhibitor’ which are substituted benzimidazoles, that suppress gastric acid secretion by specific inhibition of the H+ K+ ATPase enzyme system, the acid (proton) pump of the gastric parietal cell.
After intravenous administration of omeprazole, the onset of the antisecretory effect occurring within two hours. A single dose of 40 mg of Omeprazole given intravenously has similar effect on intragastric acidity over a 24-hour period as repeated oral dosing with 20 mg once daily. Although the plasma half-life of omeprazole is very short (50 minutes), the antisecretory effect lasts longer due to prolonged binding to the parietal H+ K+ ATPase enzyme. When the drug is discontinued, secretory activity returns gradually over 3 to 5 days. The inhibitory effect of omeprazole on acid secretion is dose-related and increases with repeated once daily dosing, reaching a plateau after four days.
Omeprazole is indicated primarily for the treatment of Zollinger- Ellison syndrome, and may also be used for the treatment of gastric ulcer, duodenal ulcer and reflux esophagitis.
Patients who are unable to take oral medication, e.g in severely ill patients with reflux esophagitis, duodenal ulcer or gastric ulcer. Omeprazole 40 mg bolus is recommended once daily for up to 5 days.
Administration
Omeprazole 40 mg I.V. injection should be administered intravenously only and should not be given by any other route.
Injection
For I.V. injection, reconstitute one sterile single dose vial of Omeprazole (Propix) Injection with 10 mL of sterile water for Injection USP (without preservative) to make 10 mL solution containing 4 mg/ mL of Omeprazole approximately.
The reconstituted solution of Omeprazole (Propix) injection having 4 mg/ mL of Omeprazole approximately is stable for 4 hours when stored in original vial in a cool place. The reconstituted solution of Omeprazole (Propix) injection should not be used if it contains visible particulate matter.
Infusion
For I.V. infusion, reconstitute one sterile single dose vial of Omeprazole (Propix) injection with 10 ml of sterile water for Injection USP (without preservative) to make 10 mL solution containing 4 mg/mL of Omeprazole approximately. Subsequently add 10 mL of reconstituted solution containing 4 mg/mL of Omeprazole approximately, to 90 mL of Sodium Chloride Injection (containing 0.9% w/v Sodium Chloride) or 90 mL of Dextrose Injection (containing 5% w/v Dextrose) or 90 mL of Mannitol Injection (containing 5% w/v of Mannitol) to make 100 mL solution of 0.4 mg/mL of Omeprazole approximately. No other solution should be used for infusion. The resultant infusion should be given intravenously over a period of 20-30 minutes. The prepared infusion solution should be used within 3 hours of preparation and any unused portion should be discarded. The infusion solution having Omeprazole should not be refrigerated.
The diluted infusion solution of Omeprazole (Propix) Injection having 0.4 mg/mL of Omeprazole approximately in infusion fluids are stable up to 4 hours when stored in a cool place and protect from light. The diluted infusion solution containing Omeprazole sodium should not be used if they contain visible particulate matter.
Check out our available products
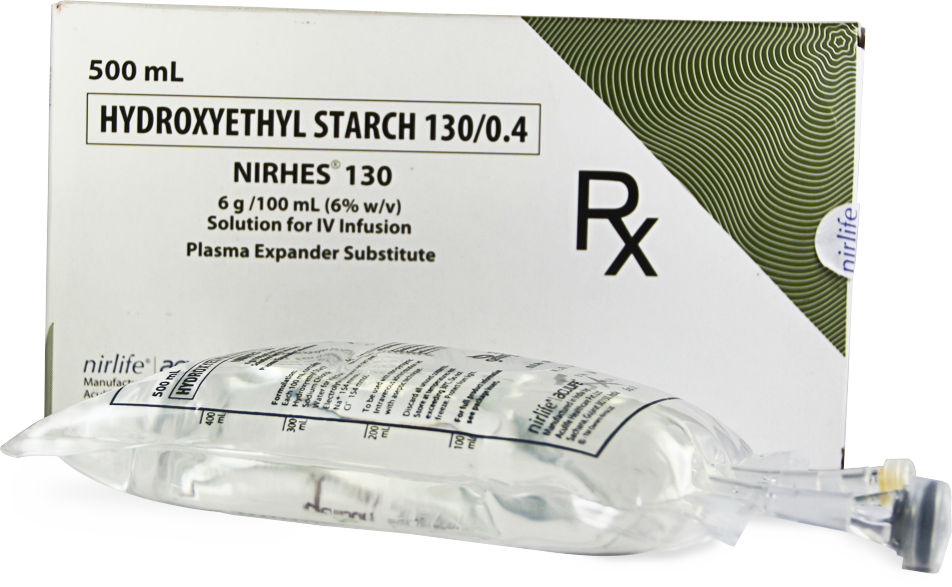
NIRHES 130
Action and Clinical Pharmacology Indications and Clinical Uses Contraindications Warning Precautions Caution Pregnancy Nursing Mothers Children Renal Impairment Adverse Reactions Symptoms and Treatment of Overdose Dosage and Administration Pediatric Dose Special Instructions...
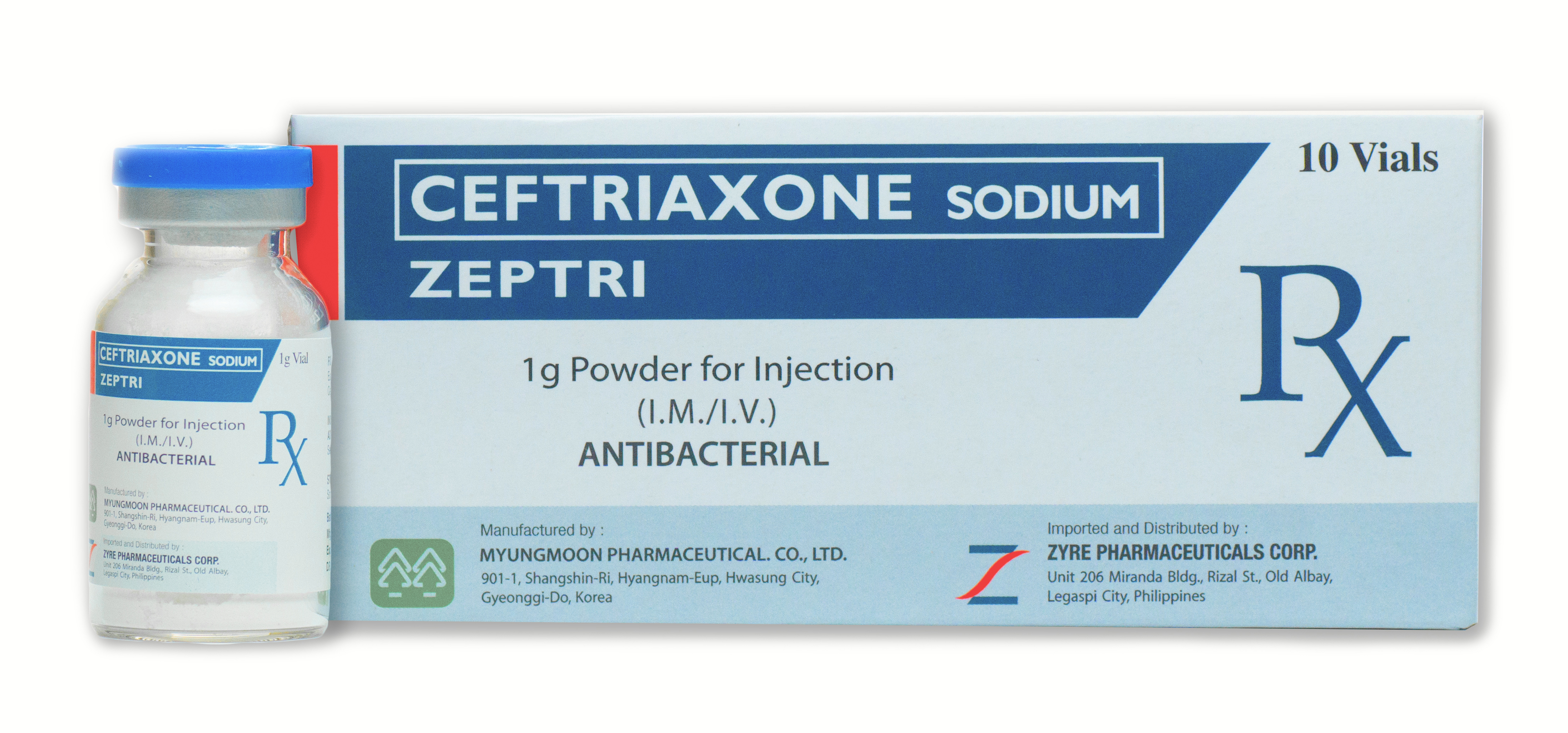
ZEPTRI
Pharmacologic Classification Therapeutic Indication Dosage Forms Bioavailability and Pharmacokinetics Dose Range Known Adverse Effects and Toxicities Special Precautions Mode Of Action Spectrum Of Activity Indications Pharmacokinetics Contraindications Adverse Reaction/Side Effects Route And...
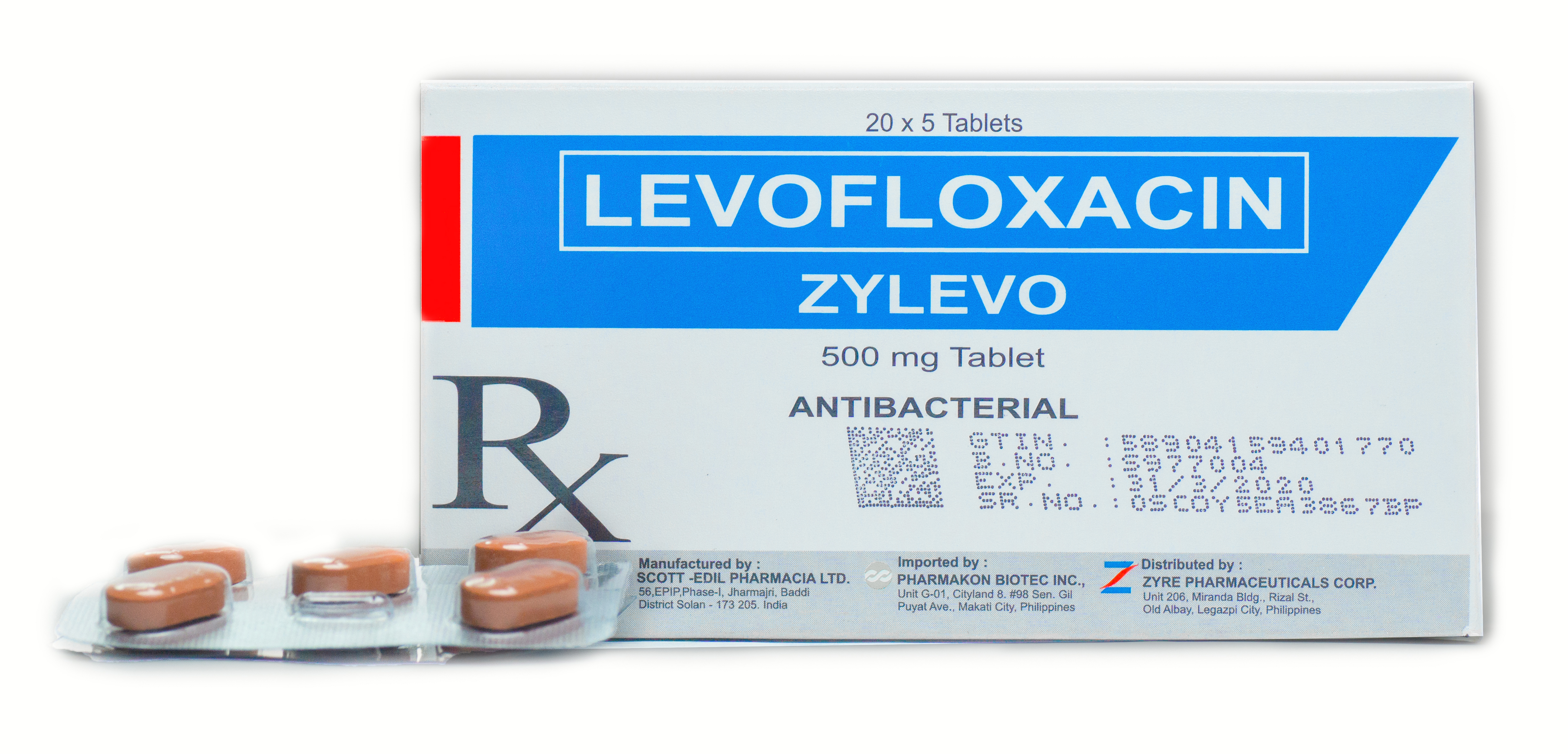
ZYLEVO
Dosage Form & Strength Pharmacological Classification Therapeutic Indication Pharmacokinetics Dosage and Administration Special Precautions Dosage Form & Strength 500 mg tablet Pharmacological Classification Levofloxacin is often bactericidal at concentrations equal to or...
Get In Touch
For questions, inquiry or to know more about our products, please feel free to call us at +63 052 742-2844 / +63 02 829-0181, email us at zyrepharma@yahoo.com or click the button below.
Contact Us