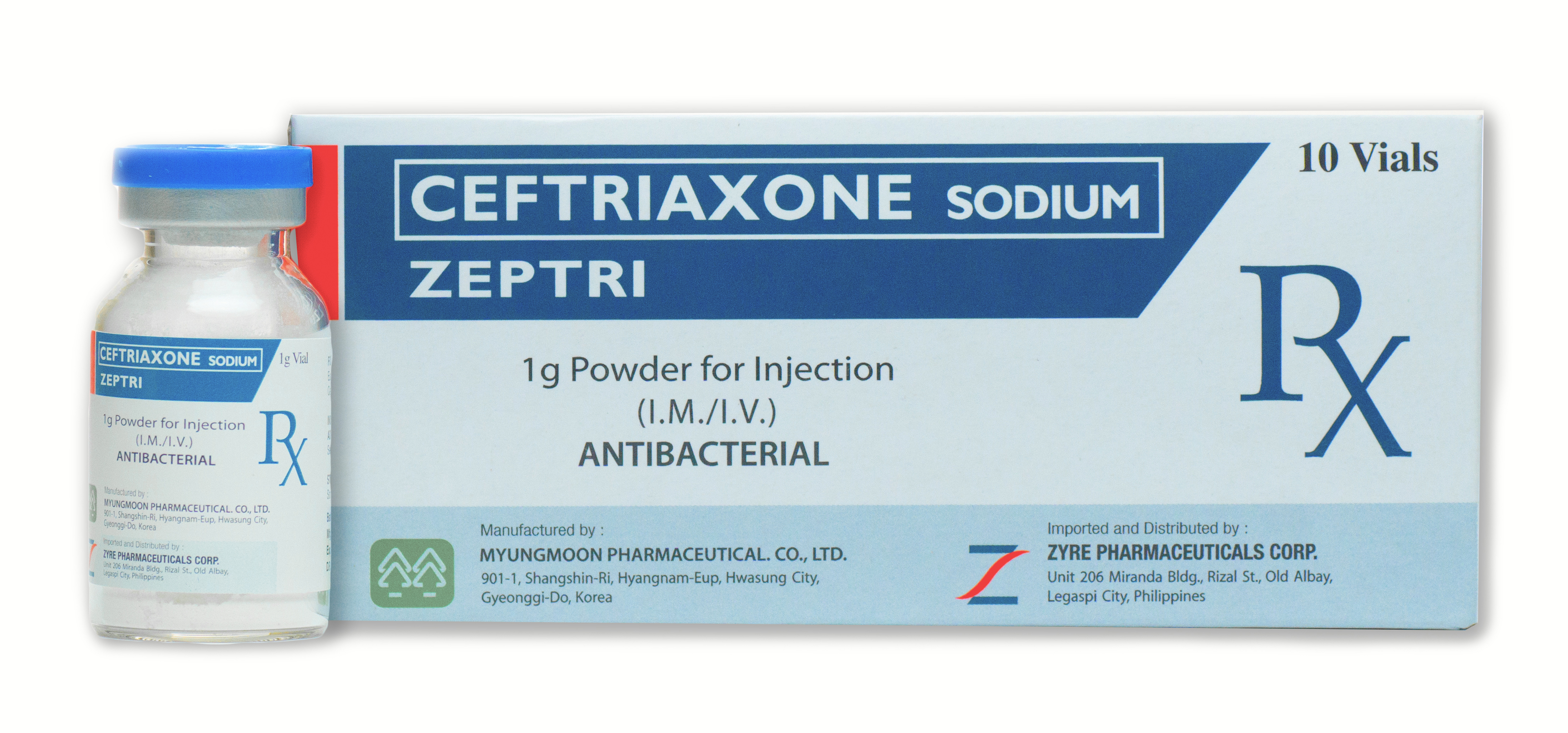
ZEPTRI
Pharmacologic Classification
Ceftriaxone is a third-generation antibiotic from the cephalosporin family of antibiotics. It is within {\displaystyle \beta } \beta -lactam family of antibiotics. Ceftriaxone selectively and irreversibly inhibits bacterial cell wall synthesis by binding to transpeptidases, also called transamidases, which are penicillin-binding proteins (PBPs) that catalyze the cross-linking of the peptidoglycan polymers forming the bacterial cell wall
Therapeutic Indication
Respiratory tract infections such as pneumonia,brochitis,etc.ear,nose ,throat infections ,renal urinary tract infections, genital infections such as gonorrhea
Dosage Forms
1g Powder for Injection (IM/IV)
Bioavailability and Pharmacokinetics
Ceftriaxone is available for administration via the intramuscular or the intravenous routes. Diluents containing calcium should not be used to reconstitute ceftriaxone and it must not be administered in intravenous lines containing other calcium-containing solutions, as a ceftriaxone-calcium precipitate could form.
Absorption: Ceftriaxone can be administered intravenously and intramuscularly, and the drug is completely absorbed.It is not available orally
Distribution: Ceftriaxone penetrates tissues and body fluids well, including cerebrospinal fluid to treat central nervous system infections. The average volume of distribution in adults is 5.8-13.5 liters.
Metabolism: 33-67% of ceftriaxone is renally excreted as unchanged drug, but no dose adjustments are required in renal impairment with dosages up to 2 grams per day. The rest is excreted in the bile as inactive compounds from hepatic and gut flora metabolism
Elimination: The average elimination half-life in healthy adults is 5.8-8.7 hours. In people with renal impairment, the average elimination half-life increases to 11.4-15.7 hours
Dose Range
1 to 2 g IV or IM once a day (or in equally divided doses twice a day)
Duration of therapy: 4 to 14 day
- Complicated infections: Longer therapy may be required.
- Infections due to Streptococcus pyogenes: At least 10 days
Comments:
- Dose and duration depend on the nature and severity of the infection.
- The total daily dose should not exceed 4 g.
Known Adverse Effects and Toxicities
The most common adverse reactions associated with ceftriaxone are changes in white blood cell counts, local reactions at site of administration, rash, and diarrhea Some less frequently reported adverse events (incidence < 1%) include phlebitis, itchiness, fever, chills, nausea, vomiting, elevations of bilirubin, elevations in creatinine, headache and dizziness
Special Precautions
Ceftriaxone and calcium -containing solutions including continuous calcium-containing infusion such as parenteral nutrition,should not be mixed or co-administered to any patient irrespective of age,even via different infusion lines at different sites Ceftriaxone is contraindicated in patients with known hypersensitivity to ceftriaxone sodium or any component of the container, other cephalosporins or penicillins Hyperbilirubinemic neonates and preterm neonates should not be treated with ceftriaxone. In vitro studies have shown that ceftriaxone can displace bilirubin from its binding to serum albumin, leading to a possible risk of bilirubin encephalopathy in these patients
Cautious treatment is required as renal insufficiency may be worsened with combination of similar compounds (other cephem antibiotics ) and diuretics such as furosemide ,etc.
Synergy between ceftriaxone and aminoglycosides has been demonstrated with many gram-negative bacilli under experimental conditions and it is of special importance in severe life-threatening infections due to microorganism such as Pseudomonas aeroginosa.
Mode Of Action
Semi-synthetic third generation cephalosporins antibiotic, having bactericidal action preferentially binds to one or more of the PCN-binding proteins (PBP) located on cell wall of susceptible organisms. This inhibits third and final stage of bacterial wall synthesis, thus killing the bacterium.
Bactericidal action of ceftriaxone demonstrates a high affinity for the B-lactam receptor protein in the cell wall of actively growing bacteria.
Resistant to B-lactamase, the structure of ceftriaxone is inherently protected against the destruction by B-lactamase and penicillinase produced by both gram (+) and gram (-) bacteria.
Spectrum Of Activity
Ceftriaxone has a degree of stability in the presence of B-lactamases, both penicillinases and cephalosporins of gram (+) and gram (-) bacteria.
Indications
Ceftriaxone is used to treat the infections caused susceptible organisms in lower respiratory tract, skin and skin structures, urinary tract, bones and joints. Also used to treat intra-abdominal infections, pelvic inflammatory disease, uncomplicated gonorrhoea, meningitis and for surgical prophylaxis.
Lower respiratory tract infections caused by Streptococcus pneumoniae, Staphylococcus aureus, Haemophilus influenza, Haemophilus para-influenzae, Klebsiella pneumoniae, Escherichia coli, Enterobacter aerogenes, Proteus mirabilis or Serratia marcescens.
Acute bacterial otitis media caused by Streptococcus pneumoniae, Haemophilus influenza (including B-lactamase producing strains) or Moraxella catarrhalis (including B-lactamase producing strains).
Skin and skin structure infections caused by Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyrogenes, Escherichia coli, Enterobacter cloacae, Klebsiella, Proteus mirabilis, Morganella morganii, Pseudomonas aeroginosa, Serratia marcescens, Acinetobacter calcoaceticus, Peptostreptococcus species.
Urinary tract infections (complicated and uncomplicated) caused by Escherichia coli, Proteus mirabilis, Proteus vulgaris, Morganella morganii or Klebsiella pneumoniae.
Uncomplicated gonorrhoea (cervical, urethral and rectal) caused by Neis. Gonorrehae, including both penicillinase-producing strains and pharyngeal gonorrhoea caused by nonpenicillinase-producing strains of Neis. gonorrhoea.
Bacterial septecimia caused by Staphylococcus aureus, Streptococcus pnuemoniae, Escherichia coli, Haemophilus influenza or Klebsiella pneumoniae.
Bone and joint infections caused by Staphylococcus aureus, Streptococcus pneumoniae, Eschericia coli, Proteus mirabilis, Klebsiella pneumoniae or Enterobacter species.
Intra-abdominal infections caused by Escherichia coli, Klebsiella pneumoniae, Bacteroides fragilis, Clostridium species (note: most strains of Clos. difficile are resistant) or Peptostreptococcus species.
Meningitis caused by Haemophilus influeanzae, Neisseria meningitidis or Streptococcus pnuemoniae. Surgical prophylaxis, the preoperative administration of a single 1g dose may reduce the incidence of postoperative infections in patients undergoing surgical procedures classified as contaminated or potentially contaminated (e.g. vaginal or abdominal hysterectomy or cholecystectomy for chronic calculous cholecystitis in high risk patients such as those over 70 years of age, with acute cholecystitis not requiring therapeutic antimicrobials, obstructive jaundice or common duct bile stones) and in surgical patients for whom at the operative site would present serious risk (e.g. during coronary artery bypass surgery).
Pharmacokinetics
Peak serum concentration: From IM route, 2-3 hours after the single dose; from IV route, within the 30minutes completion of single dose infusion.
Distribution in tissue: widely distributed throughout the body including gallbladder, lungs, bone, bile, CSF (diffuses into CSF at higher concentrations when the meninges are inflamed); crosses placenta, reaches amniotic fluid and milk.
Protein binding: 85-95%
Contraindications
- Hypersensitivity to cephalosporins and penicillin.
- Pseudo-membranous colitis has been reported with nearly all-antibacterial agents, including Ceftriaxone and may range in severity from mild to life-threatening.
- Although transient elevations of BUN and serum creatinine have been observed at the recommended dosage the nephrotoxic potential of Ceftriaxone is similar to that of other cephalosporins.
Adverse Reaction/Side Effects
GI: diarrhea (2-4%), abdominal cramps and pseudo-membranous colitis (2-4%).
Gyne: genital pruritus, moniliasis.
Others: pin in duration at IM injection site, phlebitis (IV site), pruritus, fever, chills, anaemia, haemolytic anaemia, prolonged PT, nausea, vomiting, dyspepsia, increased alkaline phosphatase, increased bilirubin, increased creatinine, urinary casts, HA, dizziness, candidiasis, vaginitis.
Route And Dosage
Ceftriaxone should be given IV or IM:
- Adults: 1-2g once a day; maximum dose: 4g/day.
- Children: 50-75mg/day, once a day or in divided doses q 12h (not to exceed 2g/day).
- Meningitis: pediatric dose, 100mg/kg/day (not to exceed 4g in divided doses q 12h with or without loading dose of 75mg/kg.
- Surgical prophylaxis: single 1g dose, ½ -2h before surgery.
- Uncomplicated gonorrhoea infections: IM: single dose, 250mg
- Therapy should be continue at least 2 days after the sign and symptoms of infection have disappeared. The usual duration of therapy is 7-14 days according to the severity of condition.
Check out our available products
NIRHES 130
Action and Clinical Pharmacology Indications and Clinical Uses Contraindications Warning Precautions Caution Pregnancy Nursing Mothers Children Renal Impairment Adverse Reactions Symptoms and Treatment of Overdose Dosage and Administration Pediatric Dose Special Instructions...

K5 CRYOLASER
PACKAGE SIZE & TYPE COMPOSITION DESCRIPTION INDICATION SUGGESTED USE CAUTION PACKAGE SIZE & TYPE 20 ml Tube COMPOSITION Dicaprylyl Ether, C12-15 Alkyl Benzoate, Cyclopentasiloxane, Zea mays (Corn) Starch, Pidobenzone, Ethylhexyl Methoxycinnamate (Octyl...

NIRPID 10
Dosage Strength & Form Descripton Pharmacology Indications Contraindications Warning Precautions Caution Adverse Reactions Dosage Infusion Rate Administration Overdosage Storage Presentation Dosage Strength & Form Dosage Strength & Form 10g/100ml (10%w/v) Fat Emulsion...
Get In Touch
For questions, inquiry or to know more about our products, please feel free to call us at +63 052 742-2844 / +63 02 829-0181, email us at zyrepharma@yahoo.com or click the button below.
Contact Us