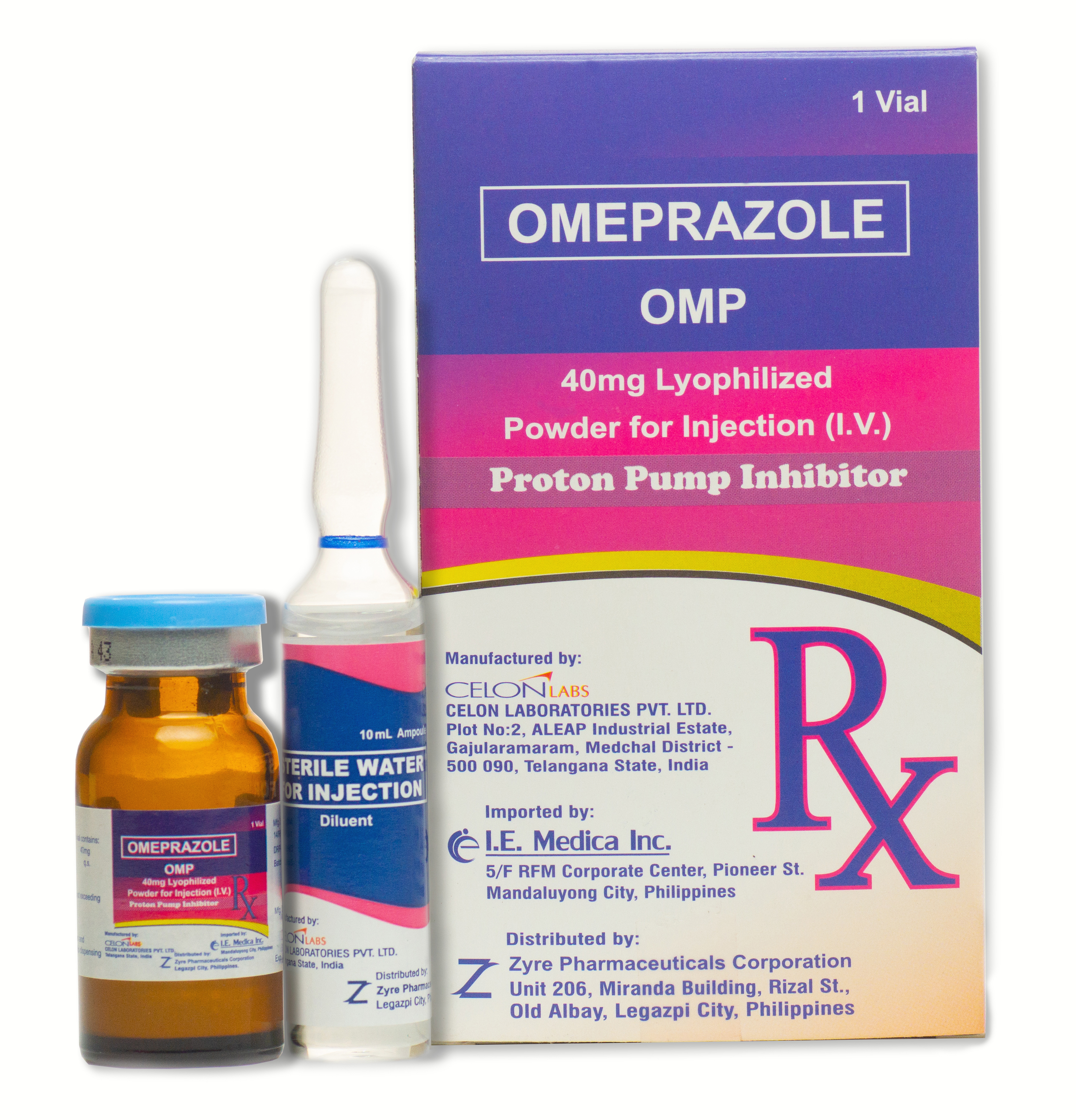
OMP
Pharmacological Information
Pharmacologic Classification
Omeprazole is activated at an acidic pH to a sulphenamide derivative that binds irreversibly to H+ K+ ATPase enzyme, an enzyme system found at the secretory surface of parietal cells. It thereby inhibits the final transport of hydrogen ions into the gastric lumen thus inhibiting acid section. Omeprazole inhibits both basal and stimulated acid secretion irrespective of the stimulus.
Therapeutic Indication
Indicated primarily for the treatment of Zollinger-Ellison syndrome and may also be used for the treatment of gastric ulcer, duodenal ulcer and reflux esophagitis.
It is indicated for prophylactic use.
Dosage Forms
40 mg vial + 10 ml diluent
Bioavailability and Pharmacokinetics
Distribution. The plasma protein binding of omeprazole is approximately 95%. The inhibition of acid secretion is related to the area under the plasma concentration time curve (AUC) but not to the actual plasma concentration at any given time.
Metabolism. Omeprazole is entirely metabolised by the cytochrome P450 system (CYP), mainly in the liver. Identified metabolites in plasma are the sulfone, the sulfide and hydroxyomeprazole. These metabolites have no significant effect on acid secretion. The average half-life of the terminal phase of the plasma concentration – time curve following IV administration of omeprazole is approximately 40 minutes; the total plasma clearance is 0.3 to 0.6L/minute. There is no change in half-life during repeated dosing
Excretion. About 80% of the metabolites are excreted in urine and the remainder in faeces. The two main urinary metabolites are hydroxyomeprazole and the corresponding carboxylic acid.
Dose Range
Dosage: in patients with duodenal ulcer, gastric ulcer or reflux oesophagitis where oral medication is inappropriate. Omeprazole (OMP) 40 mg once daily is recommended for up to 5 days. In patients with Zollinger-Ellison syndrome the recommended initial dose of Omeprazole (OMP) given intravenously is 60 mg daily. Higher daily doses may be required and the dose should be adjusted individually. when doses exceed 60 mg daily, the dose should be divided and given twice daily.
Known Adverse Effects and Toxicities
The adverse effects reported most frequently with Omeprazole have been gastrointestinal disturbances, in particular diarrhea, constipation, abdominal pain, nausea and vomiting and flatulence. Skin rash pruritus and urticaria have been reported, usually resolving after discontinuation of treatment. Central nervous system disordered reported so far include paresthesia, dizzines, light-headedness and feeling faint, all of these resolved on cessation of therapy. Other adverse events reported rarely include arthralgia and myalgia, blurred vision, taste disturbances, blood disorders including agranulocytosis, leucopenia.
Special Precautions
Precautions: When gastric ulcer is suspected, the possibility of malignancy should be excluded before treatment with Omeprazole (OMP) is instituted, as treatment may alleviate symptoms and delay diagnosis.
Drug Interactions: The absorption of some medicines might be altered due to decreased intragastric acidity. Thus it can be predicted that the absoprtion of ketoconazole and itraconazole can decrease during omeprazole treatment, as it dose during treatment with other acid secretion inhibitors or antacids. As omperazole (OMP) is metabolized in the liver through cytochrome P450, it can prolong the elimination of the diazepam, phenytoin, warfarin and other vitamin K antagonists, which are all in part substrates for this enzyme.
Pharmacological Actions
Omeprazole belongs to a class of antisecretory compounds, known as “proton pump inhibitors” which are substituted benzimidazoles, that suppress gastric acid secretion by specific inhibition of the H+ K+ ATPase enzyme system, the acid (proton) pump of the gastric parietal cell.
After intravenous administration of omeprazole, the onset of the anti-secretory effect occurs within one hour, with the maximum effect occurring within two hours. A single dose of 40 mg of Omeprazole given intravenously has similar effect on intragastric acidity over a 24-hour period as repeated oral dosing with 20 mg once daily. Although the plasma half-life of omeprazole is very short (50 minutes), the anti-secretory effect lasts longer due to prolonged binding to the parietal H+ K+ ATPase enzyme. When the drug is discontinued, secretory activity returns gradually, over 3 to 5 days. The inhibitory effect of omeprazole on acid secretion is dose-related and increases with repeated once daily dosing, reaching a plateau after 4 days.
Mechanism Of Action
Omeprazole is activated at an acidic pH to a sulphonamide derivative that binds irreversibly to H+ K+ ATPase enzyme, an enzyme system found at the secretory surface of the parietal cells, it thereby inhibits the final transport of hydrogen ions (via exchange with potassium ions) into the gastric lumen thus inhibiting acid secretion. Omeprazole inhibits both basal and stimulated acid secretion irrespective of the stimulus.
Pharmacokenetic Profile
The apparent volume of distribution of omeprazole in healthy subjects and in patients with renal insufficiency is almost similar. The volume is distribution is slightly decreased in the elderly and in patients with hepatic insufficiency. The plasma protein binding of omeprazole is about 95%. The average half-life of the terminal phase of the plasma concentration time curve following intravenous administration of omeprazole is approximately 40 minutes. Omeprazole is completely metabolized by the cytochrome P450 system, mainly in the liver. No metabolite has been found to have any effect on gastric acid secretion. Almost 80% of an intravenous dose of omeprazole is excreted as metabolites in the urine, and the remainder is found in the faeces, primarily originating from biliary secretion.
Clinical Information
Indications
Omeprazole (OMP) is indicated primarily for the treatment of Zollinger-Ellison syndrome, and may also be used for the treatment of gastric ulcer, duodenal ulcer and reflux oesophagitis.
Dosage and Administration
Patients who are unable to take oral medication, e.g. in severely ill patients with reflex oesophagitis, duodenal ulcer or gastric ulcer. Omeprazole 40 mg bolus is recommended once daily for up to 5 days.
Administration
Omeprazole 40 mg IV injection should be administered IV only and should not be given by any other route.
Injection
For IV injection, reconstitute one sterile single dose vial of omeprazole (OMP) injection with 10 ml of sterile water for injection USP (with our preservative) to make 10 ml solution containing 4mg /ml of omeprazole approximately.
The reconstituted solution of OMP injection having 4mg/ml of omeprazole approximately is stable for 4 hours when stored in original vial in a cool place. The reconstituted solution of OMP injection should not be used if it contains visible particulate matter.
Infusion
For IV infusion, reconstitute one sterile single dose vial of omeprazole sodium (OMP) injection with 10 ml of sterile water for injection USP (with our preservative) to make 10 ml solution containing 4mg/ml of omeprazole approximately. Subsequently add 10 ml of reconstituted solution containing 4 mg/ml of omeprazole approximately, to 90 ml of Sodium Chloride Injection (containing 0.9% w/v Sodium Chloride) or 90 ml of Dextrose Injection (containing 5% w/v Dextrose) or 90 ml of Mannitol Injection (containing 5% w/v of Mannitol) to make 100 ml solution of 0.4 mg/ml of omeprazole approximately. No other solution should be used in infusion. The resultant infusion should be given intravenously over a period of 20-30 minutes. The prepared infusion solution should be used within 3 hours of preparation and any unused portion should be discarded. The infusion solution having omeprazole should not be refrigerated. The diluted infusion solution of omeprazole (OMP) injection having 0.4 mg/ml of omeprazole (OMP) approximately in infusion fluids are stable up to 4 hrs when stored in a cool place and protected from light. The diluted infusion solution containing omeprazole sodium (OMP) should not be used if they contain visible particulate matter.
Use In Children
The safety and effectiveness of omeprazole sodium (OMP) Injection have not been established.
Contraindications
Patients who are hypersensitive to omeprazole sodium (OMP) injection or any of the inactive ingredients of omeprazole sodium (OMP) injection are contraindicated to use omeprazole intravenous injection.
Use In Patients With Impaired Liver Function
Dosage reduction may be required in patients with impaired liver function as omeprazole sodium (OMP) injection is extensively metabolized in the liver and its elimination rate is prolonged in such patients when compared to normal persons. The daily dose of 10 – 20 mg is recommended in patients with
Check out our available products
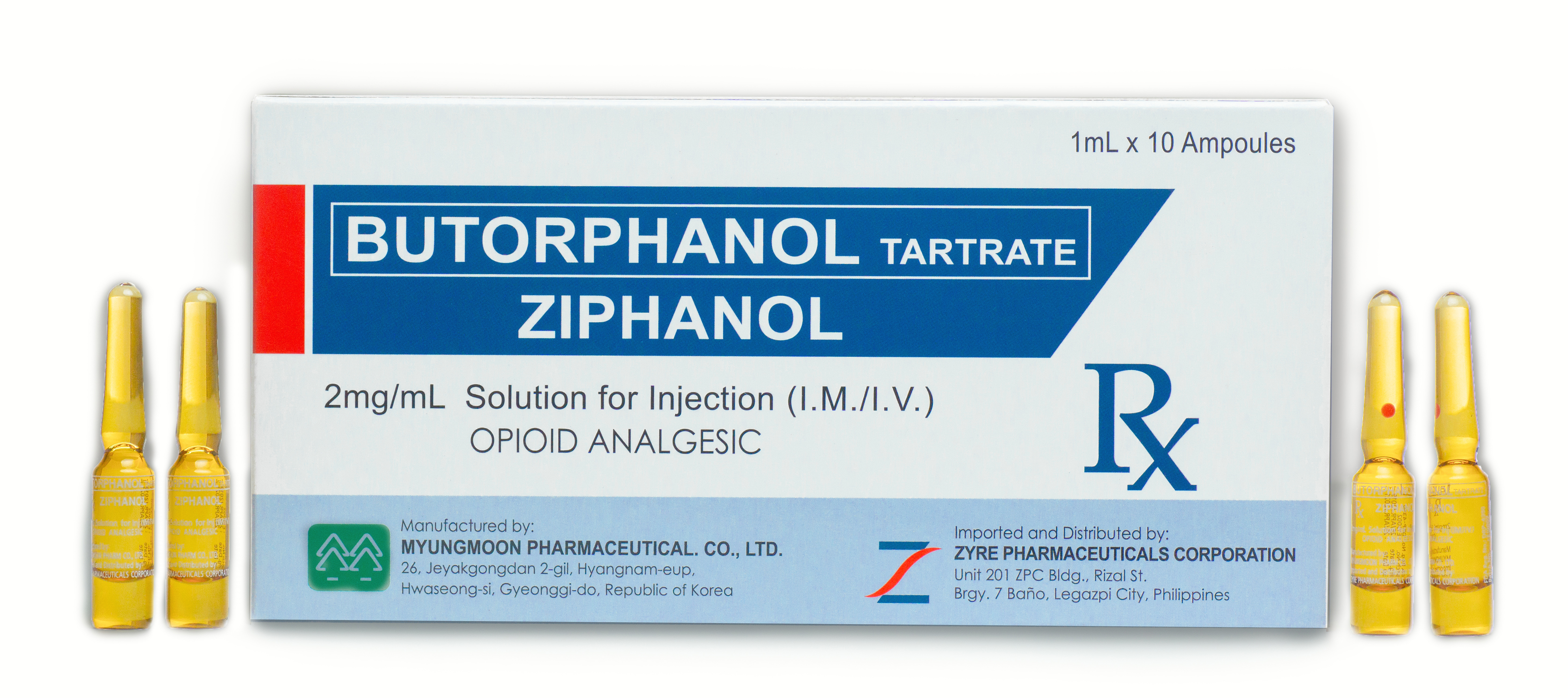
ZIPHANOL
Therapeutic Indication Dosage Forms Bioavailability and Pharmacokinetics Dose Range Known Adverse Effects and Toxicities Special Precautions Therapeutic Indication Therapeutic Indication Moderate to severe pain of any form, particularly visceral pain; perioperative analgesia....

Z-FIX
Dosage Form & Strength Pharmacological Classification Therapeutic Indication Pharmacokinetics Dosage and Administration Special Precautions Dosage Form & Strength 100 mg/ 5 mL Powder for Oral Suspension Pharmacological Classification Cefixime is bactericidal and...

K5 LIPOGEL
PACKAGE SIZE & TYPE COMPOSITION DESCRIPTION SUGGESTED USE CAUTION PACKAGE SIZE & TYPE 40 ml Tube COMPOSITION Dicaprylyl Ether, C12-15 Alkyl Benzoate, Cyclopentasiloxane, Zea mays (Corn) Starch, Pidobenzone, Ethylhexyl Methoxycinnamate, (Octyl Methoxycinnamate),...
Get In Touch
For questions, inquiry or to know more about our products, please feel free to call us at +63 052 742-2844 / +63 02 829-0181, email us at zyrepharma@yahoo.com or click the button below.
Contact Us