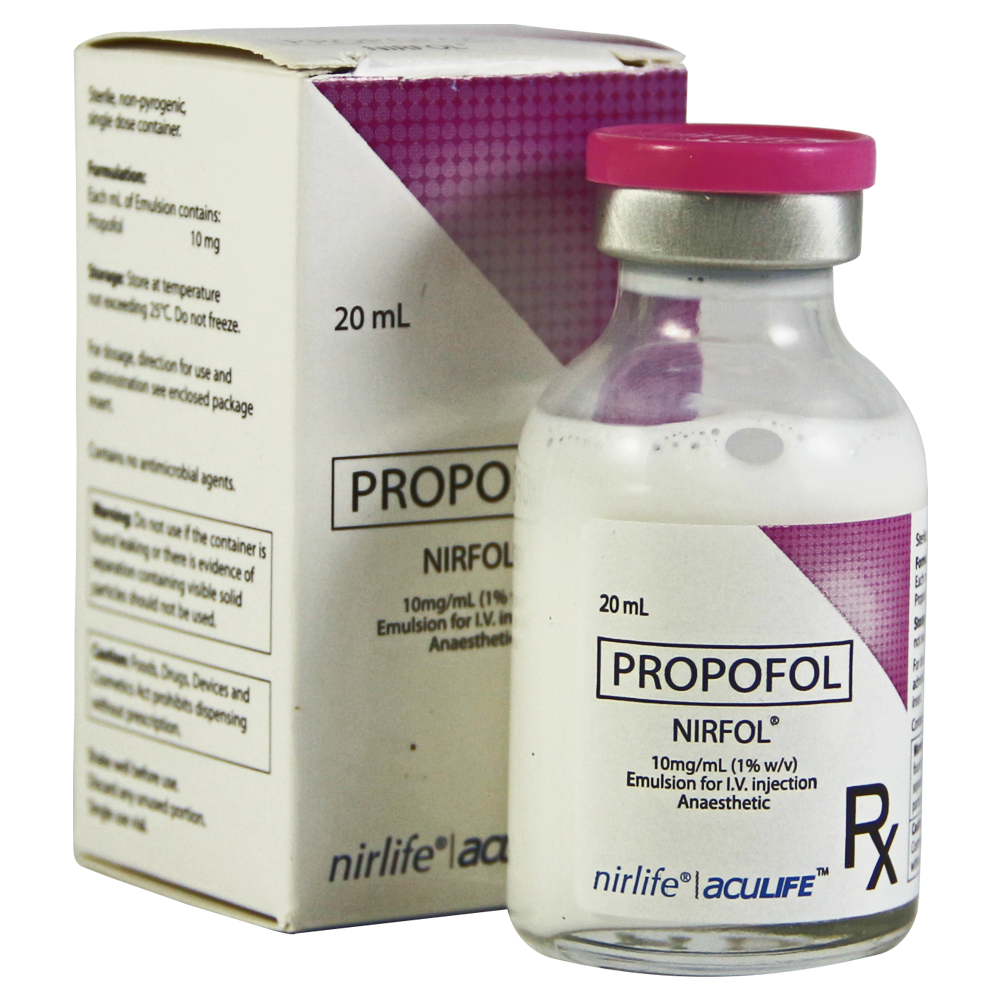
NIRFOL
Dosage Strength & Form
10mg/ml (1% w/v) Emulsion for Injection (I.V.)
Descripton
Propofol (NIRFOL) injectable Emulsion is a sterile,non-pyrogenic,oil-in-water emulsion containing 10mg/ml of propofol suitable for intravenous administration
Pharmacokinetics
Propofol (NIRFOL) is well distributed in a three compartment linear model with compartments representing the plasma, rapidly equilibrating tissues, and slowly equilibrating tissues. The first phase is characterized by a very rapid distribution (half life 2-4 min) followed by rapid elimination (half life 30-60 min.)and a slower final phase, representative of redistribution of propofol from poorly perfused tissues. Propofol is extensively distributed and rapidly cleared from the body (total body clearance 1.5-2 lit/min)Clearance occurs by the metabolic processes,mainly in the liver, to form inactive conjugates of propofol and its corresponding quinol, which are excreted in urine. When propofol is used to maintain anaesthesia, the blood concentrations, asymptomatically approach the steady state value for the given administration rate.
Indications
Propofol is a short-acting anaesthetic given intravenously for the induction and maintenance of general anaesthesia.It is also used for sedation in adult patients undergoing diagnostic procedures in those undergoing surgery in conjunction with local or regional anaesthesia, and in ventilated adult patients under intensive care.
Precautions
Propofol should not be given to patients known to be allergic to it. Propofol should be used with caution in patients with hypovolemia, epilepsy, or lipid metabolism disorders,and in the elderly. Since there have been reports of delayed convulsions associated with ease of propofol it is recommended that special care should be taken when propofol is used for day-care surgery. When used in patients with increased intacranial pressure it should be given slowly to avoid substantial decreases in mean arterial pressure and a resultant decrease in cerebral perfusion pressure. It is also recommended that propofol should not be used with ECT.
Drug Interaction
The use of propofol with other CNS depressants including those used in premedication, may increase the sedative,anaesthetic and cardio-respiratory depressant effects of propofol. It is recommended that propofol is given after opioids so that the dose of propofol can be carefully titrated against the response. The dosage of propofol should be reduced if used with nitrous oxide or halogenated anaesthetics. Although propofol does not potentiate the effects neuromuscular blockers, bradycardia and asystole have occurred after use of propofol with atracurium or suxamethonium.
Adverse Effects
Adverse effects include pain on injection especially if the injection is into a small vein. Apnoea may be frequent;apnoea lasting longer than 60 seconds has been reported to occur in 12% of patients. Cardiovascular effects include a reduction in blood pressure and bradycardia. There Lave reports of convulsions aid involuntary movements. Fever has occurred. Discoloration of urine has been reported following prolonged used. Anaphylactic like reactions have been reported. Nausea, vomiting and headache may occur during recovery.
Dosage & Administration
Propofol is available as a 1 or 2% emulsion.The 1% emulsion may be given by intravenous injection or infusion,but the 2% emulsion is for infusion only .Infusions and injections should be prepared using aseptic techniques.
Intensive Care Unit Sedation
Propofol (NIRFOL) Injectable Emulsion should be individualized according to the patient’s condition and response, blood lipid profile and vital signs. For incubated, mechanically ventilated adult patients, Intensive Care Unit (ICU) sedation should be initiated slowly/with a continuous infusions in order to titrate to desired clinical effect and minimize hypotension. When indicated,initiation of sedation should begin at 5ug/kg/min (0.3mg/kg/h) the infusion rate should be increased by increments of 5 to 10ug/kg/min (0.3-0.6mg/kg/h) until the desired level of sedation is achieved. A minimum period of 5 to 10 minutes between adjustments should be allowed for onset of peak drug effect. Mast adult patients require maintenance rates of 5 to 50ug/kg/min (0.3 to 3mh/kg/h)or higher. Dosages of NIRFOL should be reduced in patients who have received large dosages of narcotics. Conversely, adequate management of pain may reduce the Propofol (NIRFOL) Injectable Emulsion dosage requirement with analgesic agents. As with other sedative medications, there is inter patient variability in dosage requirements may change with time.
Evaluation of level of sedation and assesstment of CNS function shld be carried out daily throughout maintenance to determine the minimum dose of Propofol (NIRFOL) injectable emulsion required for sedation.
Bolus administration of 10 or 20mg should be used to rapidly increase in patients where hypotension is not likely to occur. Patients w/ compromised myocardial function, intravascular volume depletion or abnormally low vascular tone(e.g.sepsis) maybe more susceptible to hypotension
Induction of General Anaesthesia
Healthy Adult Less than 55 yrs of age 40mg every 10 seconds until induction onset(1.5 to 2.5mg/kg)Elderly Debilitated or ASA III/ IV Paients :20mg every 10 seconds until induction onset (1 to 1.5mg/kg) CardiacAnaesthesia:20mg every 10 seconds until induction onset (0.5 to 1.5mg/kg)Neurosurgical patients:20mg every 10 secons until induction onset(1 to 2mg/kg) Pediatric patients : healthy from 3 yrs to 16 yrs of age: 2.5 to 3.5mg/kg administered over 20-30 seconds
Maintenance of General Anaesthesia
Healthy Adult Less than 55 yrs of age :100 to 200 ug/kg/min (6 to 12mg/kg/h),(0.1 to 0.2mg/kg/min) Elderly Debilitated,ASAIII/IV patients: 50 to 100ug/kg/min (3 to 6mg/kg/h).(0.05 to 0.1 mg/kg/min)
Cardiac Anaesthesia
Primary Propofol (NIRFOL) Injectable Emulsion w/ secondary opioid:100-150ug/kg/min Low Dose Propofol (NIRFOL) Injectable Emulsion w/ primary opioid :50-100ug/kg/min.Neurosurgical patients:100-200 ug/kg/min (6to 12mg/kg/h).Pediatric patients:healthy from 2 months of age to 16 yrs of age:125 to 300ug/kg/min(7.5 to 18mg/kg/h)Following the first half hour o maintenance,if clinical signs of light anaesthesia are not present the infusion rate should be decreased Intermittent bolus Healthy Adult less than 55 yrs of ageLIncrements of 20 to 50 mg as needed
Initiation of MAC Sedation
Healthy Adult Less than 55 yrs of age:Slow infusion or slow injection techniques are recommended to avoid apnea or hypotension .Most patients require an infusion of 100 to 150ug/kg/min (6 to 9 mg/kg/h) for 3 to 5 mins or slow injection of 0.5 mg/kg over 3 to 5 mins followed immediately by a maintenance infusion. Elderly, delibilitated neurosurgical,or ASA III/IV
Patients: Most patients require dosages similar to healthy adults.Rapid boluses are to be avoided.
Maintenance of MAC Sedation
Healthy Adult Less than 55 yrs of age : A variable rate infusion technique is preferable over an intermittent bolus technique. Most patients require an infusion of 25 to 75 ug/kg/min (1.5 to 4.5 mg/kg/h) or incremental bolus doses of 10mg or 20mg. In elderly debilitated,neurosurgical or ASA III/IV patients. Most patients require 80% of the usual adult dose. A rapid (single or repeated) bolus dose should not be used. Initiation and maintenance of ICU Sedation in intubated, mechanically ventilated. Adult patients because of the residual effects of previous anaesthetic or sedation agents,in most patients the initial infusion should be 5 ug/kg/min(0.3mg/kg/h)for at least 5 mins.Subsequent increments of 5 gto 10ug/kg/min(0.3 to 0.6mg/kg/h)over 5 to 10 mins may be used until desired clinical effect is achieved. Maintenance rates of 5 to 50ug/kg/min (0.3 to 3mg/kg/h) or higher may be required.Evaluation of clinical effect and assessment of CNS function should be carried out daily throughout maintenance to determine the maximum dose of Propofol required for sedation. The tubing and any unused portions of Propofol should be discarded after 12 hrs because Propofol (NIRFOL) contains no preservatives and is capable of supporting growth of microorganisms.
Compatibility and Stability
Propofol (NIRFOL) Injectable Emulsion should not be mixed w/ other therapeutic agents prior to administration .Dilution prior to Administration: Propofol (NIRFOL) Injectable emulsion is provided as a ready to use formulation. However should dilution be necessary. It should only be diluted with 5% Dextrose Injection, and it should not be diluted to a concentration less than 2mg/ml because it is an emulsion.In diluted form it has been shown to be more stable when in contact w/ glass than w/ plastic (95% potency after 2 hrs of running infusion in plastic.
Administration
Compatibility of Propofol (NIRFOL) Injectable Emulsion w/ the co administration of blood /serum/plasma has been established.
Handling Procedures
Do not use if there is evidence of separation of the phases of the emulsion.Rare cases of self-administration of Propofol Injectable Emulsion by health care professionals have been reported, including some fatalities.
Availability
Propofol (NIRFOL) Injectable Emulsion is available in ready to use in USP Type II clear glass bottle in 20ml and 50ml infusion vials containing 10mg/ml of propofol. Propofol undergoes oxidative degradation in the presence of oxygen ,and is therefore packaged under nitrogen to eliminate this degradation path
Overdosage
It is likely to cause cardiorespiratory depression .Respiratory depression should be treated by artificial ventilation w/ oxygen.Cardiovascular depression may require repositioning of the patient by raising the patient’s legs,increasing the flow rates of intravenous fluids and administering pressor agents and /or anticholinergic agents.
Storage
Store at temperature not exceeding 25 C. Do not freeze.Shake well before use.
Check out our available products
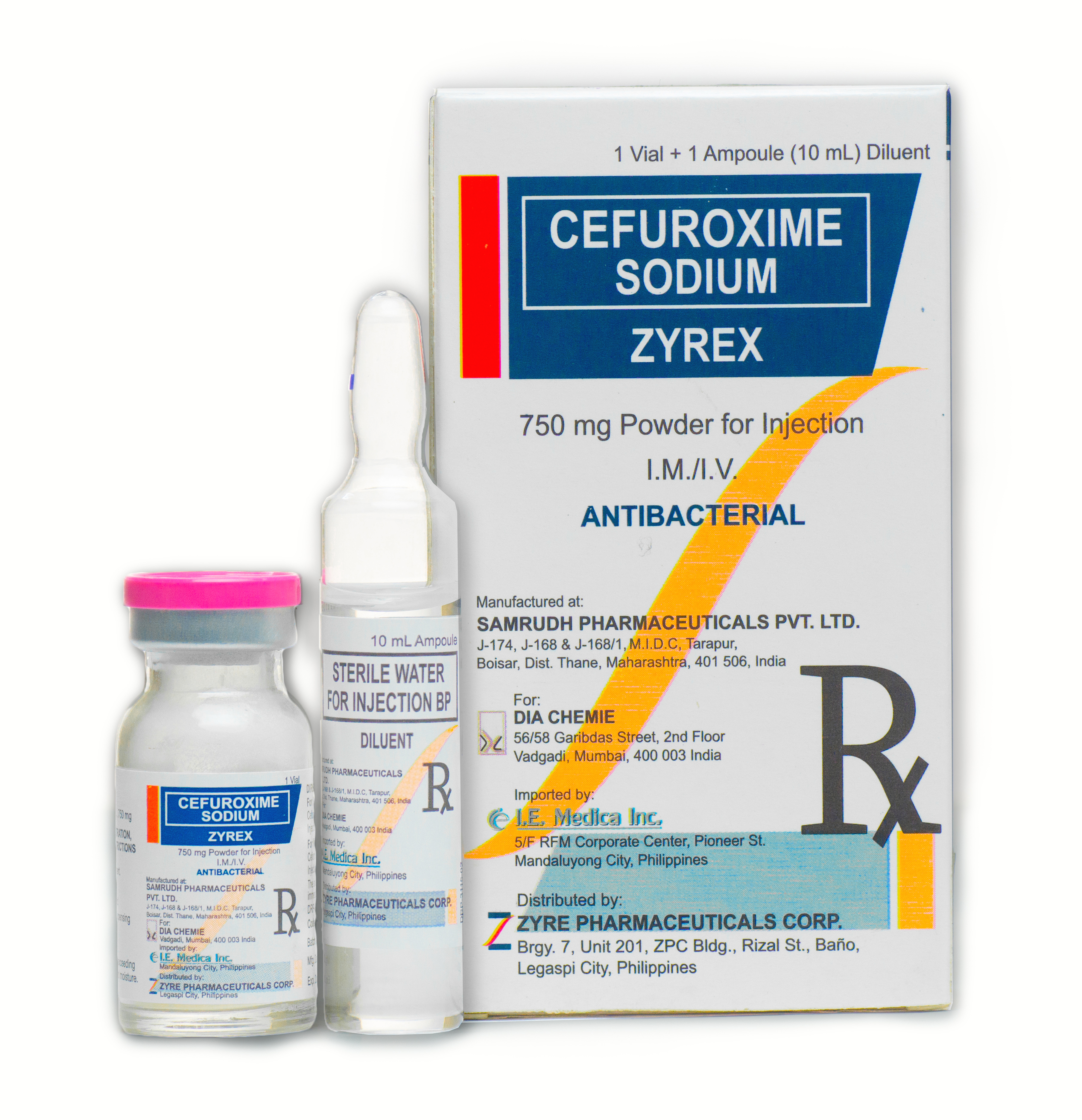
ZYREX
Cefuroxime is a semi-synthetic, broad-spectrum, second generation cephalosporins antibiotic forparenteral administration. Solutions of cefuroxime range in color from light yellow to amber, depending on the concentration and diluents used. The pH of...
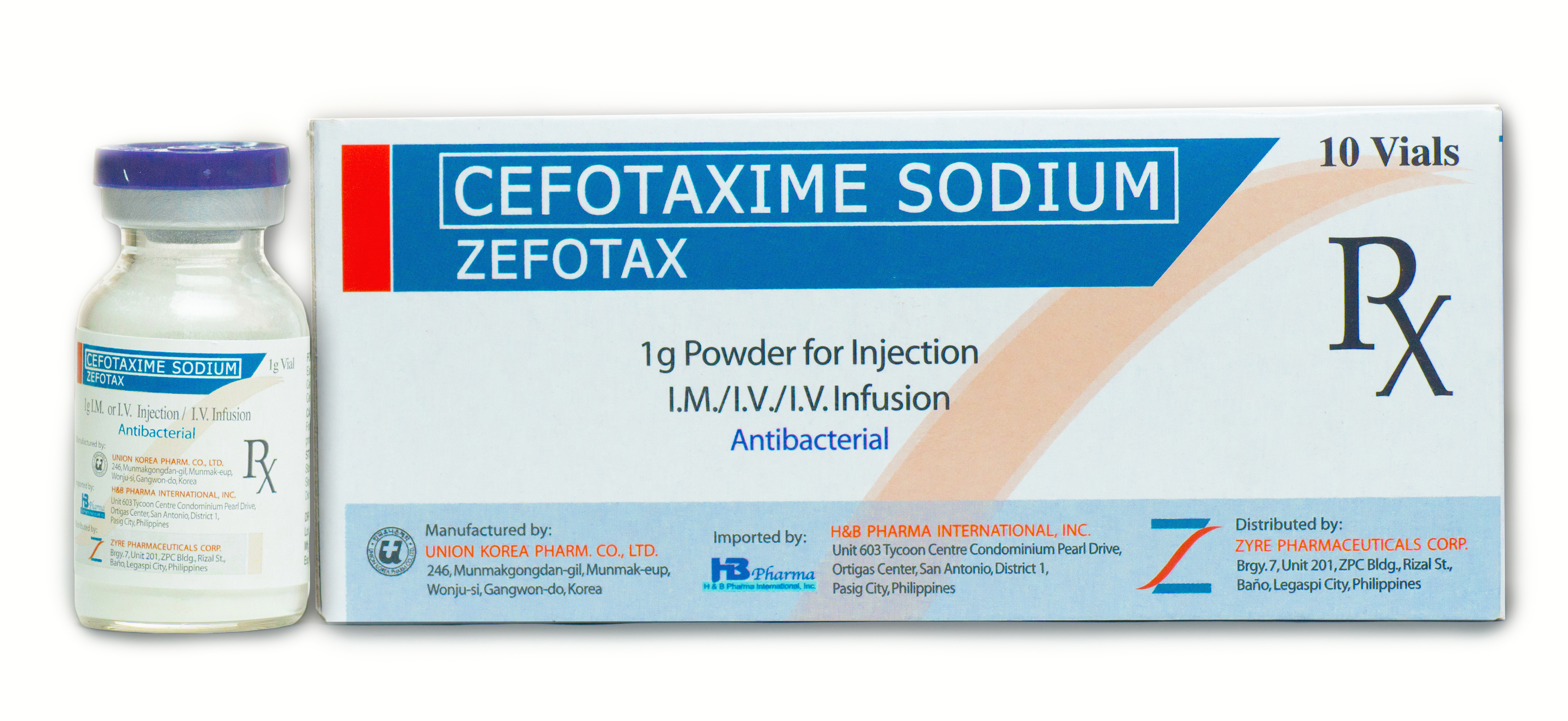
ZEFOTAX
Dosage Form & Strength Therapeutic Indication Dosage and Administration Special Precautions Dosage Form & Strength 500 mg/ 1g Powder for Injection (I.V/I.M) Therapeutic Indication Used in the treatment of infections due to...

DROXIEM
Pharmacologic Classification Therapeutic Indication Dosage Forms Bioavailability and Pharmacokinetics Dose Range Known Adverse Effects and Toxicities Special Precautions Pharmacologic Classification Pharmacologic Classification Hydroxyzine competes with histamine for binding at H1-receptor sites on...
Get In Touch
For questions, inquiry or to know more about our products, please feel free to call us at +63 052 742-2844 / +63 02 829-0181, email us at zyrepharma@yahoo.com or click the button below.
Contact Us